Cat Genetic Profile – Hypertrophic Cardiomyopathy (HCM)

Written by Dr. David W. Silversides, DVM
In cats as in humans, hypertrophic cardiomyopathy (HCM) is one of the most common heart diseases seen clinically (Kittleson 2015). In both species, symptoms include a pathological thickening of the heart muscle wall (hypertrophy), arrhythmias, obstructive embolisms, shortness of breath, exercise intolerance, heart failure and sudden cardiac death. Studies in humans have revealed that HCM is caused by structural and functional problems at the level of heart muscle fibers. The disease presents as a genetic disease with autosomal dominant transmission; indeed, over 1400 mutations in at least 11 different genes have been implicated in the human disease.
Clinical Symptoms in the Maine Coon and Ragdoll

HCM is seen in many cat breeds, but is particularly associated with the Maine Coon and Ragdoll breeds. There is considerable variation in the time of onset of clinical signs, which can occur anywhere between 1 and 4 years of age or older. Initial clinical signs of the disease can include a heart murmur when listening with a stethoscope and evident cardiac hypertrophy via an ultrasound exam. Symptoms may be mild, moderate or severe, can stabilize or can progress. In general, affected male cats tend to develop HCM symptoms that are more severe and of earlier onset than affected female cats.
The HCM Genetic Mutation
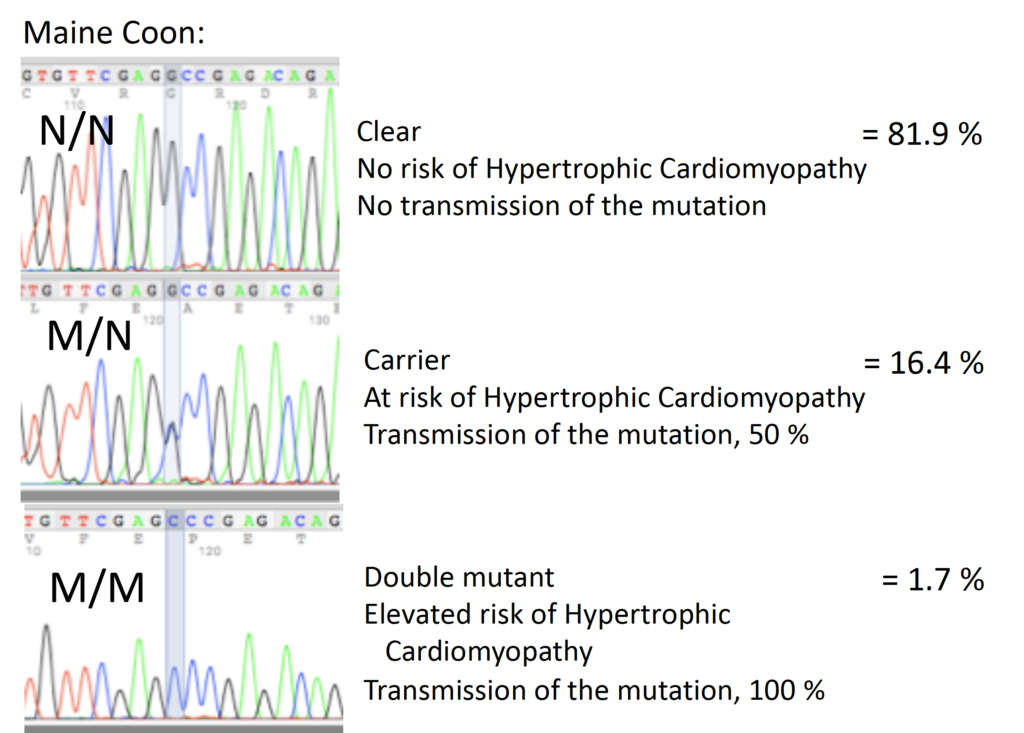
Knowledge of the human disease was used to identify two separate mutations in the feline MYBPC3 gene. The MYBPC3 gene codes for a heart muscle regulatory protein that helps to control the speed and force of heart contraction; mutations in the MYBP3 gene are a major contributing factor in HCM in humans. In the Maine Coon, the mutation is a simple substitution of one genetic letter (base) for another: a normal G is replaced by a C (Meurs 2005). In initial survey studies, 31% of Maine Coon cats were carriers of this mutation, while 3% of Maine Coons were double mutant (homozygote dominant) (Fries 2008). For the Maine Coon samples submitted to Labgenvet to date, 81.9% were N/N (clear), 16.4% were M/N (carriers) and 1.7% were M/M (double mutant).
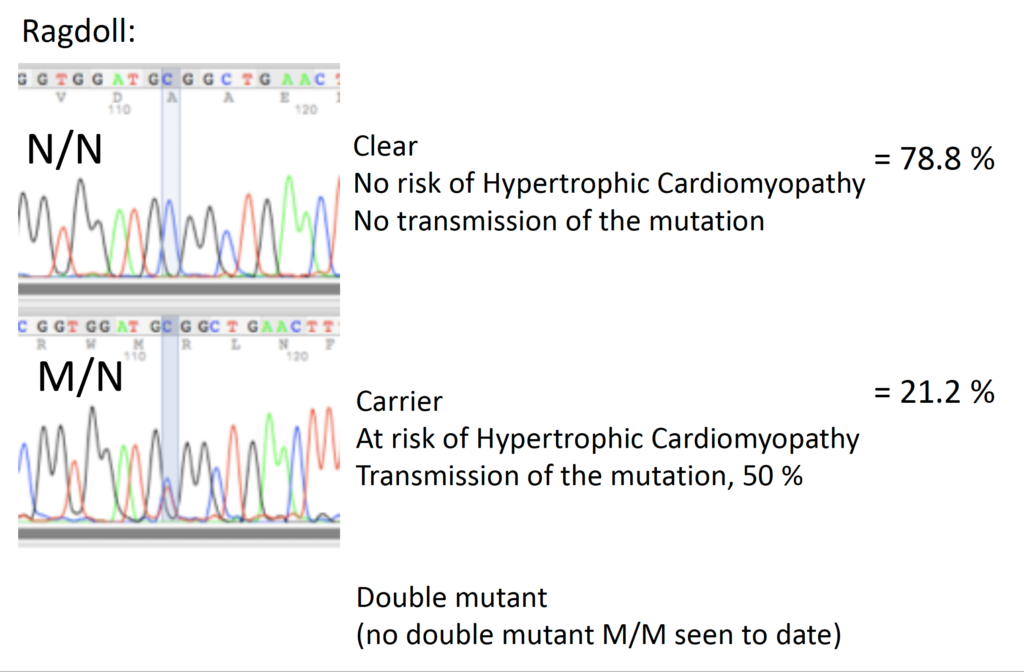
In the Ragdoll, at another place in the MYBPC3 gene, a C is replaced by a T (Meurs 2007). Frequencies of this mutation were shown to be similar to what was seen in the Maine Coon (Borgeat 2014). For the Ragdoll samples submitted to Labgenvet to date, 78.8% were N/N (clear) and 21.2% were M/N (carriers). No M/M (double mutant) animals were detected to date.
Complicated Genetics
In both cat breeds, being double mutant (M/M) results in a high chance (high “penetration” of over 50%) of a cat developing HCM. When the animal has only one mutated copy of the gene (M/N), there is a much lower chance (low penetration of about 8%) of a cat developing HCM. Indeed, there are cases of HCM in both Maine Coons, Ragdolls and other breeds of cats that do not carry the known mutations within the MYBPC3 gene. Taken together, this suggests that although HCM is a dominant genetic disease, as in the case of the human disease, the genetics in the cat are not simple. There are undoubtedly other causative mutations that have not as yet been identified, as well as other “modifier” genes that affect the function of the known mutations.
Identifying Carrier Animals

What accounts for the relatively high frequency of the known mutations for HCM in both Maine Coon and Ragdoll breeds of cats is not well understood. The fact that symptoms of HCM often occur after animals have been used for reproduction means that there has been little natural selection against the mutations. Also, the low penetration of disease within the carrier animal (M/N) makes these animals difficult to identify clinically. The identification of the mutations that contribute to HCM in Maine Coon and Ragdoll breeds allows carrier animals to be identified more precisely, and DNA tests are helping to reduce the mutation frequencies over time. Because of the relatively high frequency of these mutations as well as the low penetration of the disease in the carrier animals (M/N), several practical breeding recommendations can be made. Breeding of double mutant animale (M/M) should be discouraged, as this maintains the mutation and the risk of disease in the population. Breeding of carrier animals (M/N) to clear animals (N/N) will help to reduce the frequency of disease in the population, and can be considered for several generations. Eventually, after several generations of genetic testing, breeding can be performed to eliminate the mutation.
References:
- Meurs KM, Sanchez X, David RM et al. (2005) A cardiac myosin binding protein C mutation in the Maine coon cat with familial hypertrophic cardiomyopathy. Hum. Mol. Genet. 14:3587-93. [pubmed/16236761]
- Meurs KM, Norgard MM et al. (2007) A substitution mutation in the myosin binding protein C gene in ragdoll hypertrophic cardiomyopathy. Genomics 90(2) :261-64. [pubmed/17521870]
- Fries R, Heaney AM, Meurs KM. (2008) Prevalence of the myosin-binding protein C mutation in Maine Coon cats. J Vet Intern Med 22:893-896. [pubmed/18498321]
- Borgeat K, Casamian-Sorrosal D, Helps C et al. (2014) Association of the myosin binding protein C3 mutation (MYBPC3 R820W) with cardiac death in a survey of 236 Ragdoll cats. J Vet Cardiol 16(2) :73-80. [pubmed/24906243]
- Kittleson MD, Meurs KM, Harris SP (2015) The genetic basis of hypertrophic cardiomyopathy in cats and humans. J Vet Cardio 17:S53-S73. [pubmed/26776594]